GLP-1 drugs have dominated biopharma headlines for two years running. Novo Nordisk and Eli Lilly are generating tens of billions off injectable semaglutide and tirzepatide, and every healthcare investor is now fixated on the same question: who takes the oral GLP-1 crown?
Structure Therapeutics (NASDAQ: GPCR) is betting on GSBR-1290 (Aleniglipron), a non-peptide small molecule GLP-1 agonist designed for once-weekly oral dosing. No injections, no fasting, no cold chain. If it works, GSBR-1290 could capture a meaningful slice of a market projected to exceed $100 billion by 2030.
"If it works" is doing a lot of heavy lifting in that sentence. To separate signal from noise, we ran a comprehensive 7-step investment due diligence on Structure Therapeutics using K-Dense Web. The platform queried 9 public data sources, generated 15 visualizations, and compiled a 52-page PDF report in a single session.
Here's the breakdown.
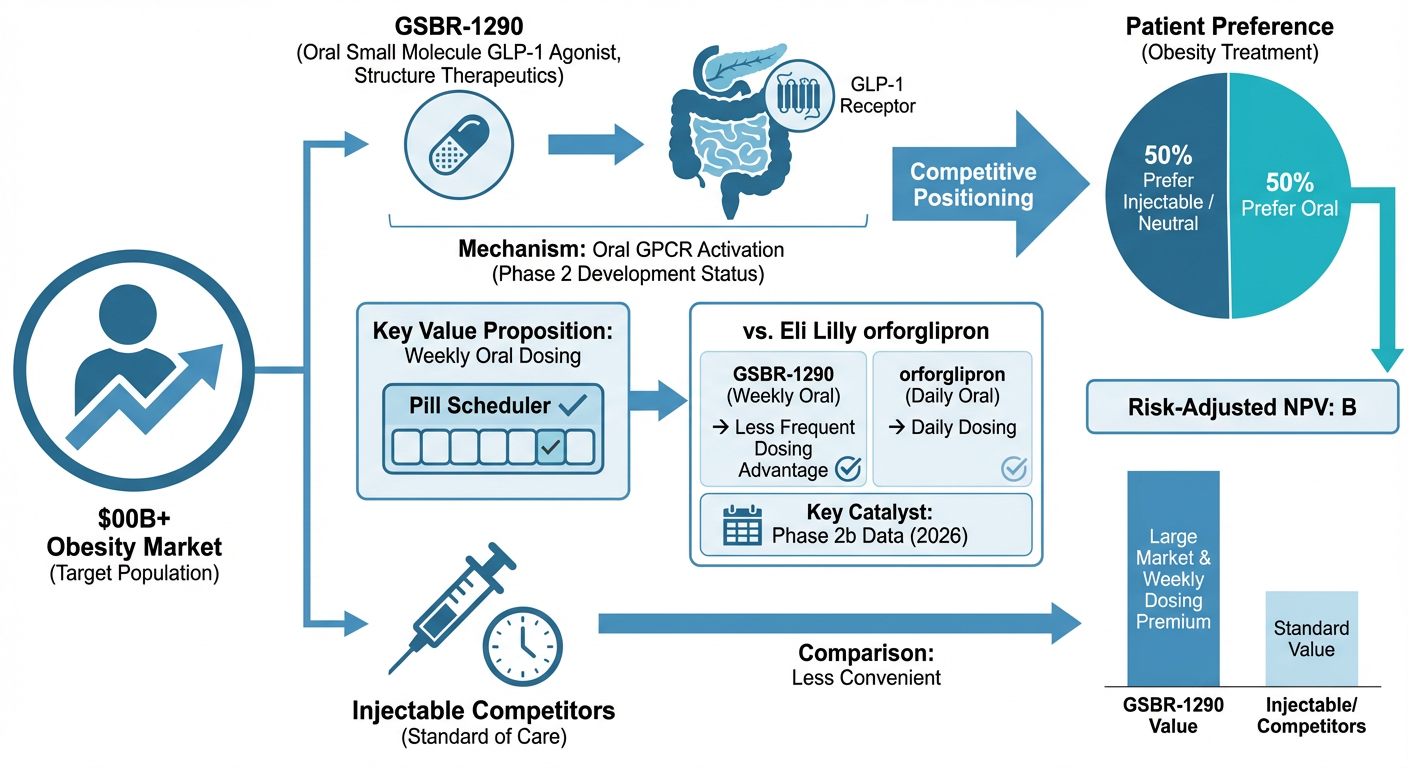 K-Dense Web generated a complete investment due diligence package for Structure Therapeutics, covering target validation, competitive intelligence, safety profiling, market sizing, IP analysis, and investment thesis construction.
K-Dense Web generated a complete investment due diligence package for Structure Therapeutics, covering target validation, competitive intelligence, safety profiling, market sizing, IP analysis, and investment thesis construction.
What K-Dense Web analyzed
The due diligence session executed a structured 7-step workflow, each step building on data from the prior analysis:
| Step | Analysis Area | Key Data Source | Key Output |
|---|---|---|---|
| 1 | Target & Scientific Validation | Open Targets, PubMed (148 articles) | GLP1R validation profile |
| 2 | Competitive Landscape Mapping | ClinicalTrials.gov (280 trials) | Competitive positioning matrix |
| 3 | Clinical Precedence & Safety | FDA FAERS (226K adverse event reports) | Safety benchmark report |
| 4 | Advanced Scientific Validation | bioRxiv, KOL analysis (38 KOLs) | MOA differentiation memo |
| 5 | Market Analysis & Sizing | CDC NHANES, CMS Part D data | TAM/SAM/SOM model |
| 6 | IP & Patent Analysis | USPTO/WIPO (23 patents) | Freedom-to-operate assessment |
| 7 | Investment Thesis & Risk | All prior data synthesized | SWOT, Porter's Five Forces, NPV |
Total data sources queried: 9 (Open Targets, PubMed, ClinicalTrials.gov, OpenFDA FAERS, bioRxiv, CDC NHANES, CMS Part D, USPTO/WIPO, SEC EDGAR)
Total outputs generated: 26 data files, 15 visualizations, 6 detailed analysis reports, and 1 compiled PDF.
The target: GLP1R is validated, but competitive
Querying the Open Targets Platform API, the analysis confirmed that GLP1R (ENSG00000112164) is one of the most validated targets in obesity and diabetes research:
| Metric | Value |
|---|---|
| Obesity association rank | #5 (score: 0.725) |
| T2DM association rank | #10 (score: 0.761) |
| Approved drugs targeting GLP1R | 15 |
| Drug-target associations | 308 |
Well-trodden path, strong validation, fierce competition. Fifteen drugs already target this receptor. For GSBR-1290 to matter, it needs to offer something genuinely different.
That difference is modality: oral, non-peptide, once-weekly.
Competitive landscape: 280 trials and counting
K-Dense Web pulled 280 active GLP-1 clinical trials from ClinicalTrials.gov and mapped the landscape by dosing modality and development phase.
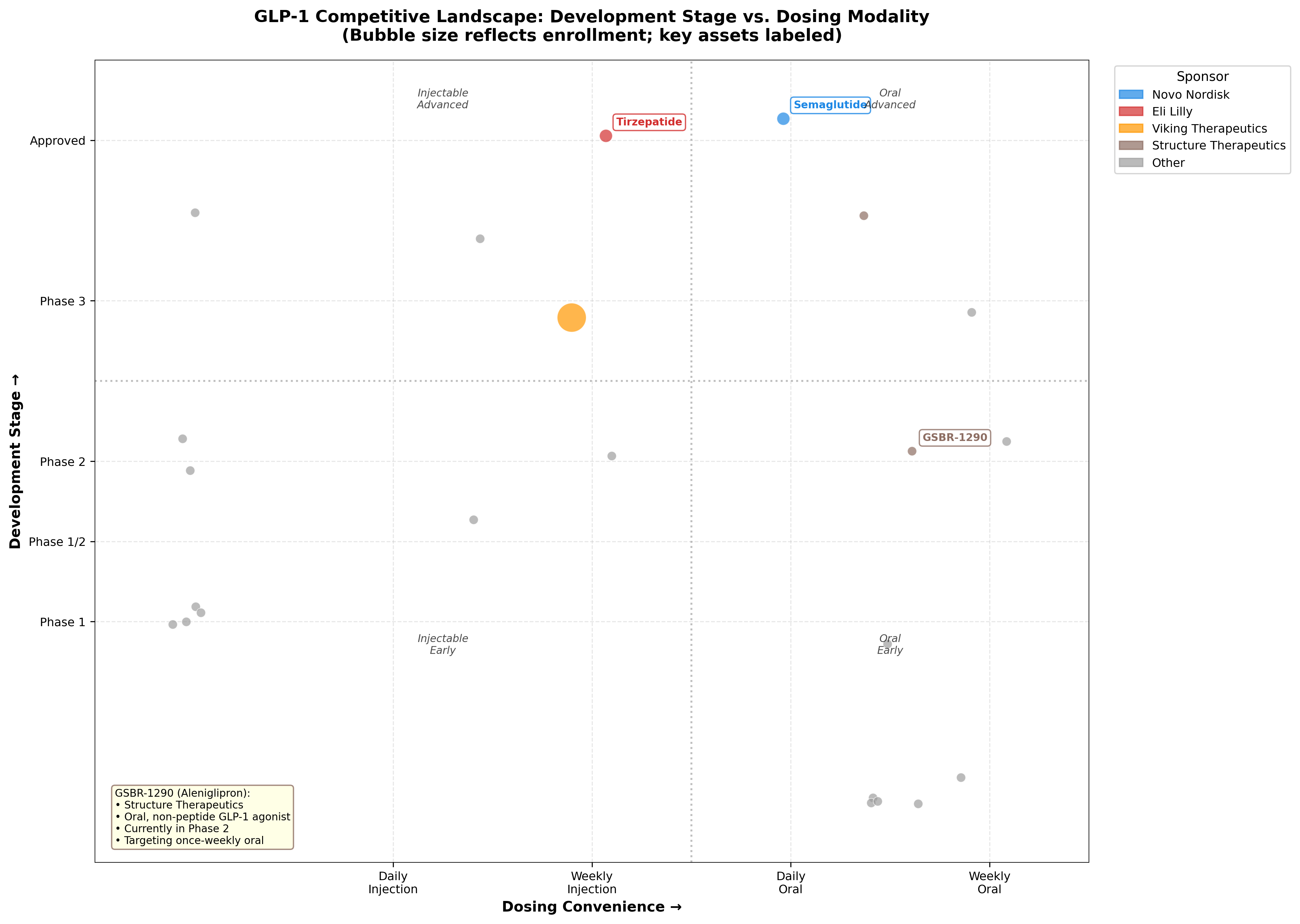 Figure 1: Competitive positioning matrix. GSBR-1290 targets the "weekly oral" quadrant, an unoccupied niche with no approved products.
Figure 1: Competitive positioning matrix. GSBR-1290 targets the "weekly oral" quadrant, an unoccupied niche with no approved products.
| Competitor | Sponsor | Active Trials | Stage | Modality |
|---|---|---|---|---|
| Semaglutide (Ozempic/Wegovy) | Novo Nordisk | 70 | Approved | Injectable weekly / Oral daily |
| Tirzepatide (Mounjaro/Zepbound) | Eli Lilly | 65 | Approved | Injectable weekly |
| Orforglipron | Eli Lilly | Phase 3 | Phase 3 | Oral daily |
| VK2735 | Viking | Phase 3 | Phase 3 | Injectable weekly |
| GSBR-1290 | Structure Therapeutics | 4 | Phase 2b | Oral weekly (target) |
No approved product currently sits in the weekly oral quadrant. That's the gap GSBR-1290 is targeting. Eli Lilly's orforglipron, while daily rather than weekly, is 12 to 18 months ahead in development and is the most direct competitive threat to watch.
What makes GSBR-1290 different: the small molecule advantage
K-Dense Web generated a mechanism-of-action differentiation memo comparing small molecule GLP-1 agonists against peptide-based competitors. The differences are structural and economic:
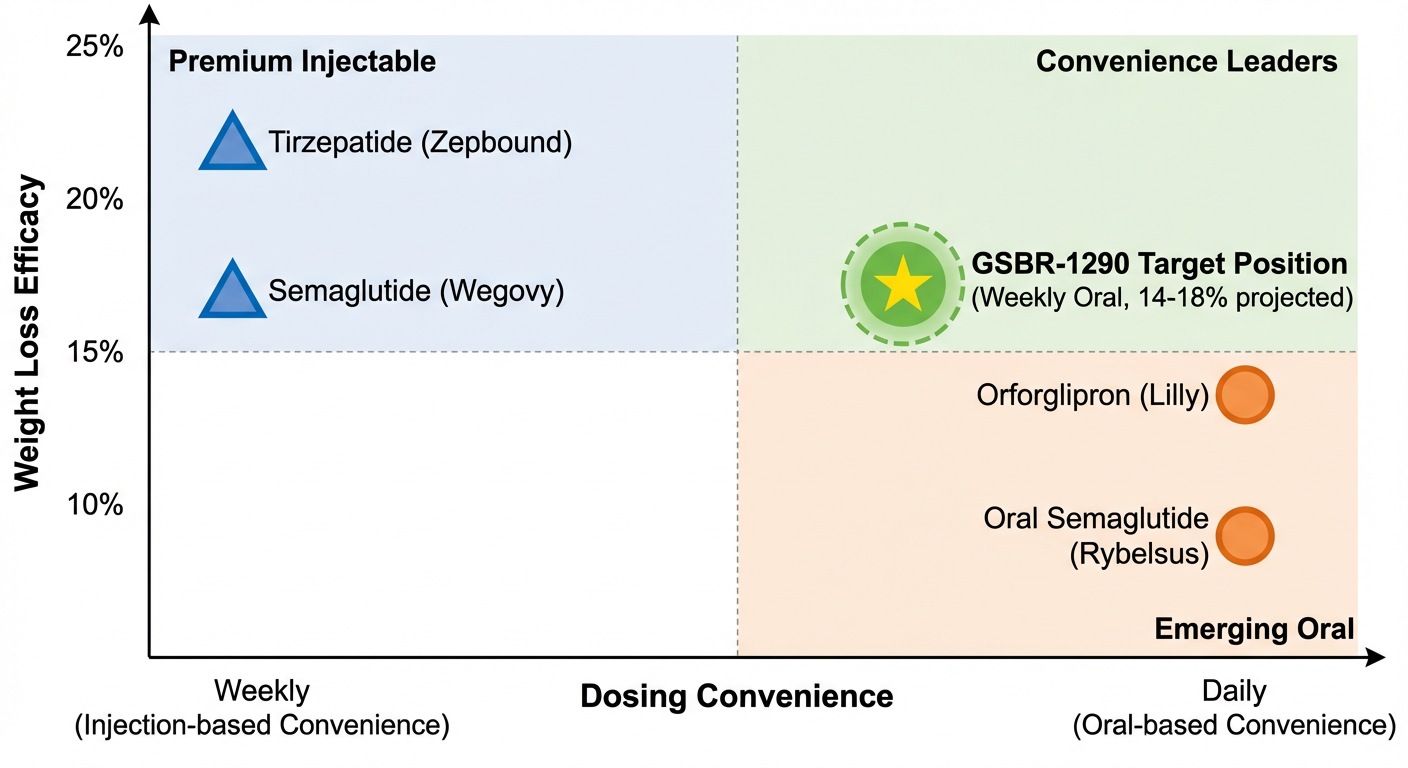 Figure 2: Competitive efficacy positioning analysis. GSBR-1290 Phase 2 data shows 11-15% weight loss at 36 weeks across doses.
Figure 2: Competitive efficacy positioning analysis. GSBR-1290 Phase 2 data shows 11-15% weight loss at 36 weeks across doses.
| Advantage | GSBR-1290 (Small Molecule) | Semaglutide / Tirzepatide (Peptide) |
|---|---|---|
| Manufacturing | Chemical synthesis (50-80% lower COGS) | Biomanufacturing / fermentation |
| Cold chain | Not required (room temperature stable) | Refrigeration required (2-8°C) |
| Fasting requirement | None | 30 min fasting for oral semaglutide |
| Oral bioavailability | Expected >30-50% | 0.4-1% (oral semaglutide with SNAC) |
| Dosing | Once weekly (target) | Daily oral or weekly injectable |
The manufacturing economics are what stand out. Chemical synthesis at scale using commodity chemicals and existing CMO infrastructure could yield 50 to 80% lower cost of goods versus peptide biologics. Eliminating cold chain requirements cuts estimated distribution costs by 15 to 25% and opens up markets where refrigeration infrastructure is thin.
The bioavailability gap is also worth noting. Oral semaglutide (Rybelsus) hits only 0.4 to 1% bioavailability and needs an absorption enhancer plus strict fasting protocols. GSBR-1290's small molecule architecture targets 30 to 50%+ without those constraints.
Safety benchmarking: class effects are real
Before any GLP-1 investment, you need to understand the safety landscape. K-Dense Web pulled 226,000+ adverse event reports from FDA FAERS and benchmarked safety signals across three approved GLP-1 agonists.
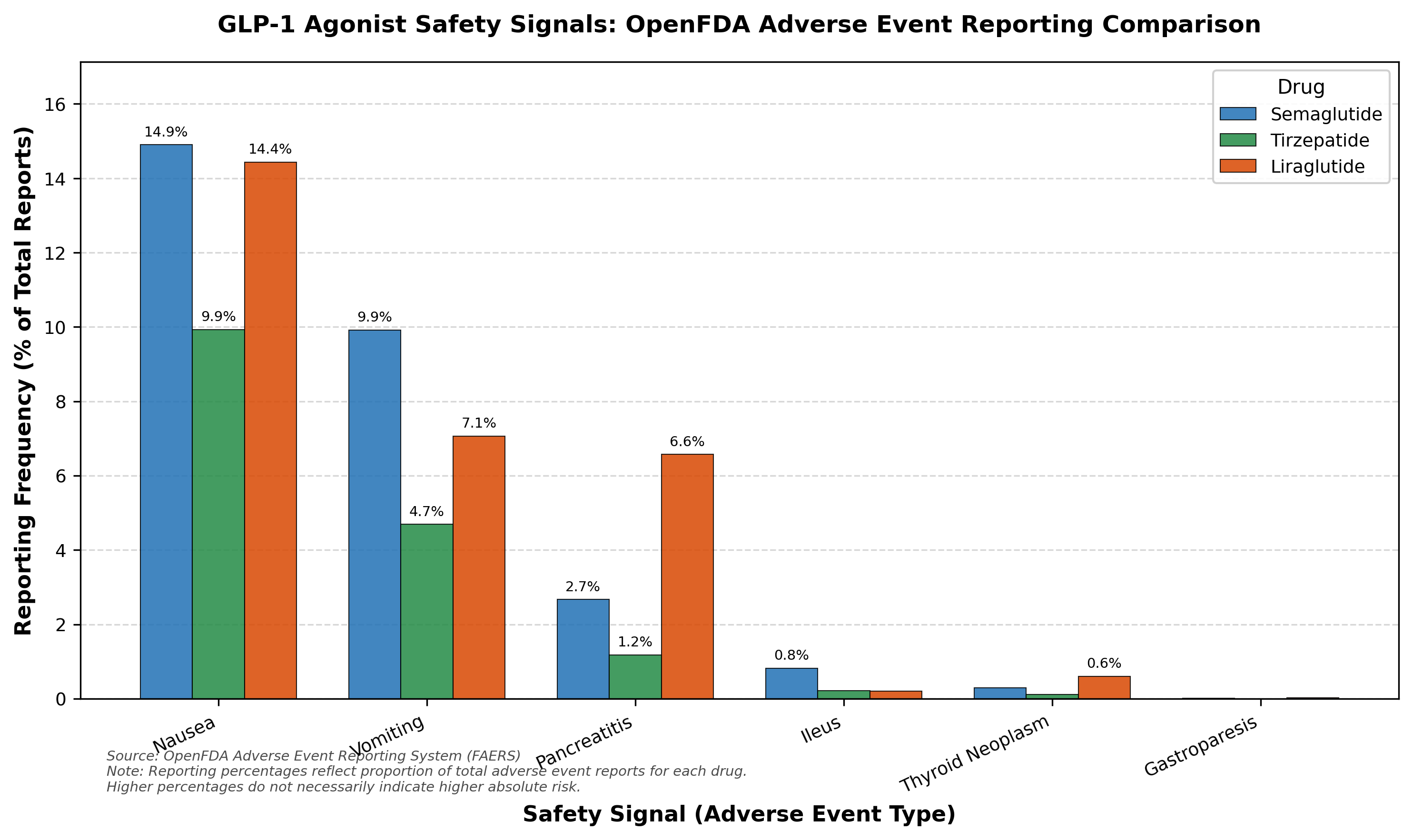 Figure 3: Safety signal comparison from FDA FAERS. GI tolerability is a universal class effect, and tirzepatide shows a somewhat more favorable profile.
Figure 3: Safety signal comparison from FDA FAERS. GI tolerability is a universal class effect, and tirzepatide shows a somewhat more favorable profile.
| Adverse Event | Semaglutide | Tirzepatide | Liraglutide |
|---|---|---|---|
| Nausea | 14.9% | 9.9% | 14.4% |
| Vomiting | 9.9% | 4.7% | 7.1% |
| Pancreatitis | 2.7% | 1.2% | 6.6% |
| Thyroid neoplasm | 0.29% | 0.11% | 0.60% |
| Ileus | 0.82% | 0.22% | 0.20% |
GI tolerability (nausea, vomiting) is a class-wide problem that requires dose titration protocols. Every GLP-1 agonist carries a thyroid C-cell tumor boxed warning from rodent studies. GSBR-1290 will face the same regulatory expectations. Whether its small molecule binding profile can achieve better tolerability remains to be demonstrated in Phase 2b.
Market sizing: a $6.5 billion peak opportunity
K-Dense Web built a bottom-up TAM/SAM/SOM model using CDC NHANES prevalence data and CMS Part D drug spending data.
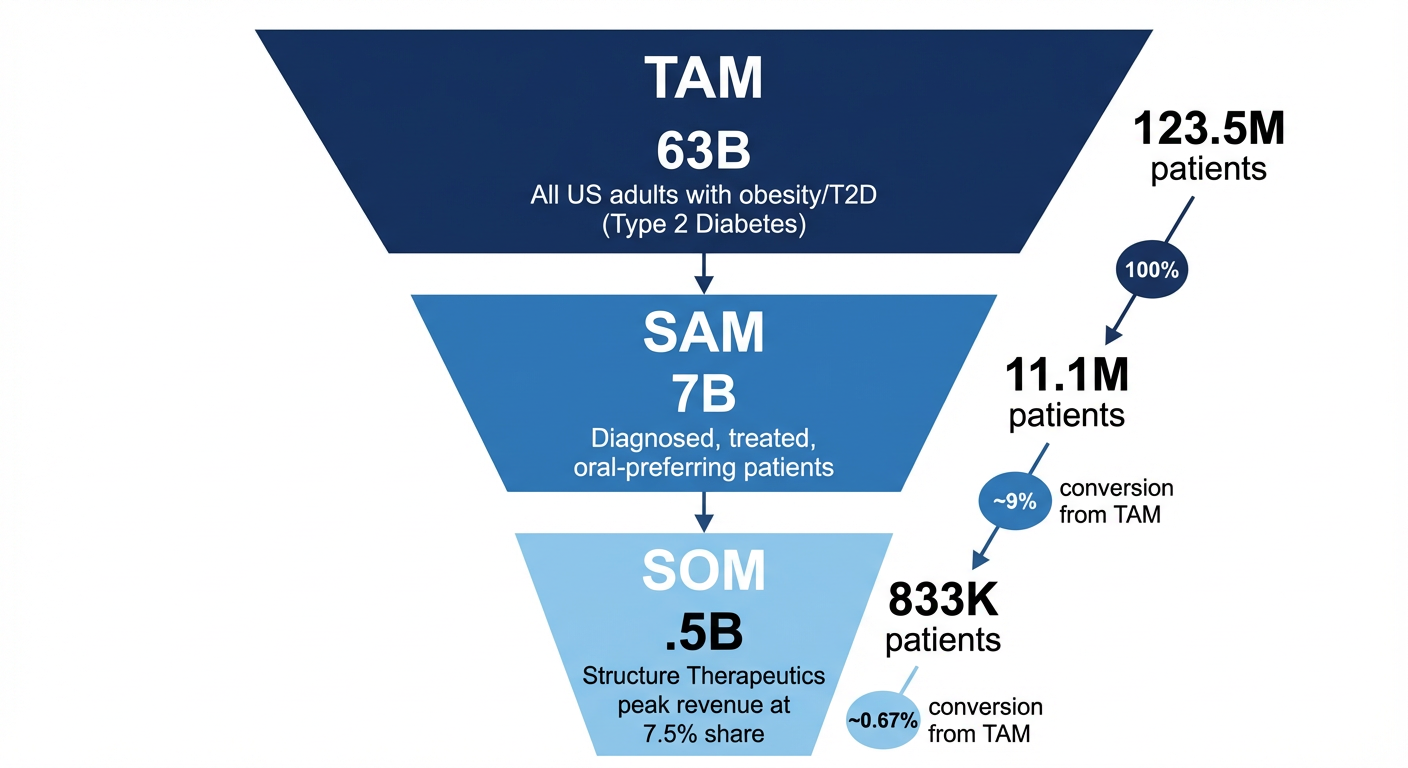 Figure 4: Market sizing funnel from total addressable market to serviceable obtainable market for GSBR-1290.
Figure 4: Market sizing funnel from total addressable market to serviceable obtainable market for GSBR-1290.
| Tier | Patients | Annual Value | Methodology |
|---|---|---|---|
| TAM | 123.5M | $963B | All US adults with obesity (42%) or T2D (11%) |
| SAM | 11.1M | $87B | Diagnosed (60%) × Treated (30%) × Oral-preferring (50%) |
| SOM (Base Case) | 834K | $6.5B | 7.5% market share at projected pricing |
The scenario analysis explored pricing and market share combinations:
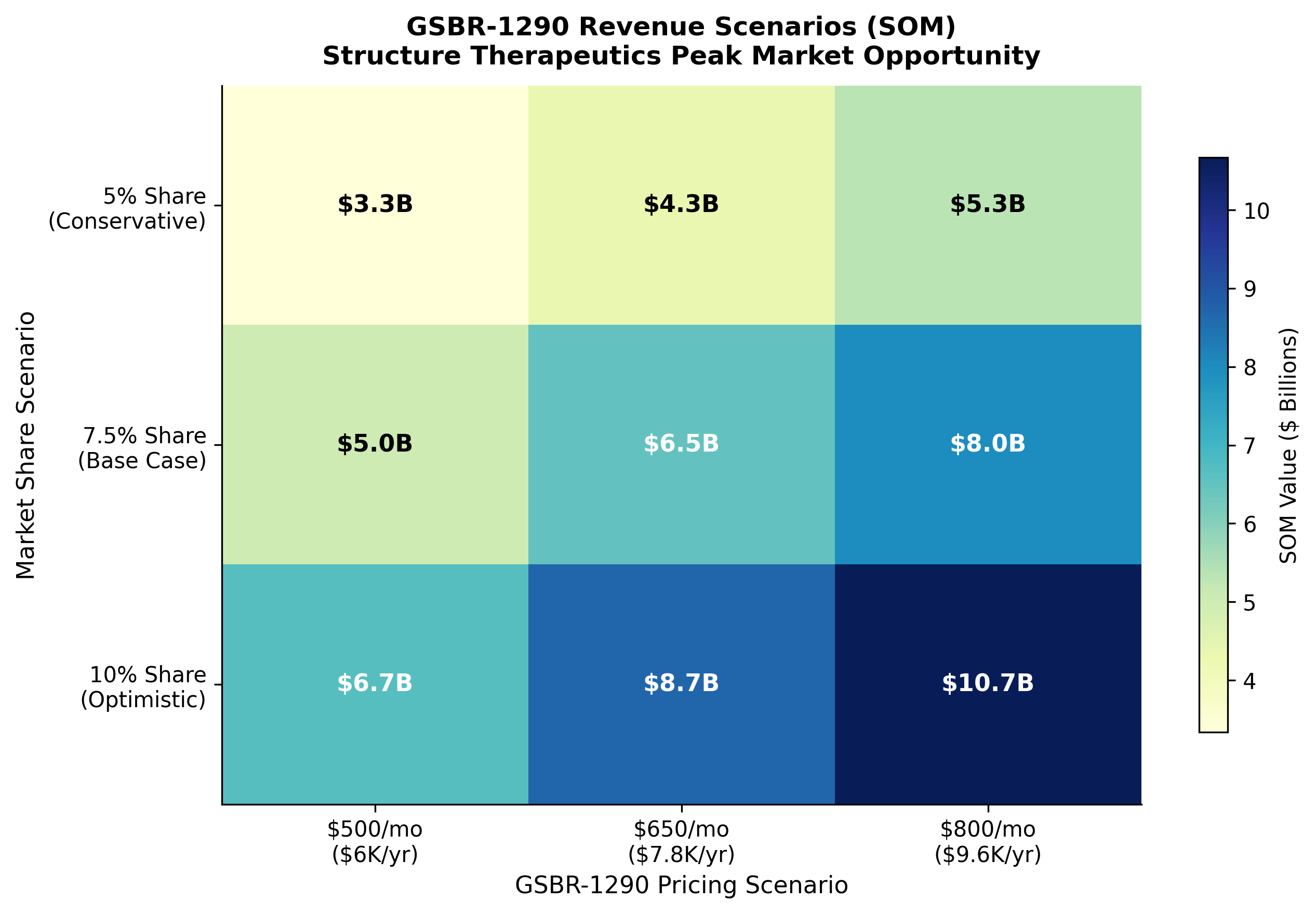 Figure 5: Revenue scenario heatmap. The base case assumes 7.5% market share at $7,800/year pricing.
Figure 5: Revenue scenario heatmap. The base case assumes 7.5% market share at $7,800/year pricing.
Pricing analysis
K-Dense Web benchmarked GSBR-1290's projected pricing against approved GLP-1 therapies:
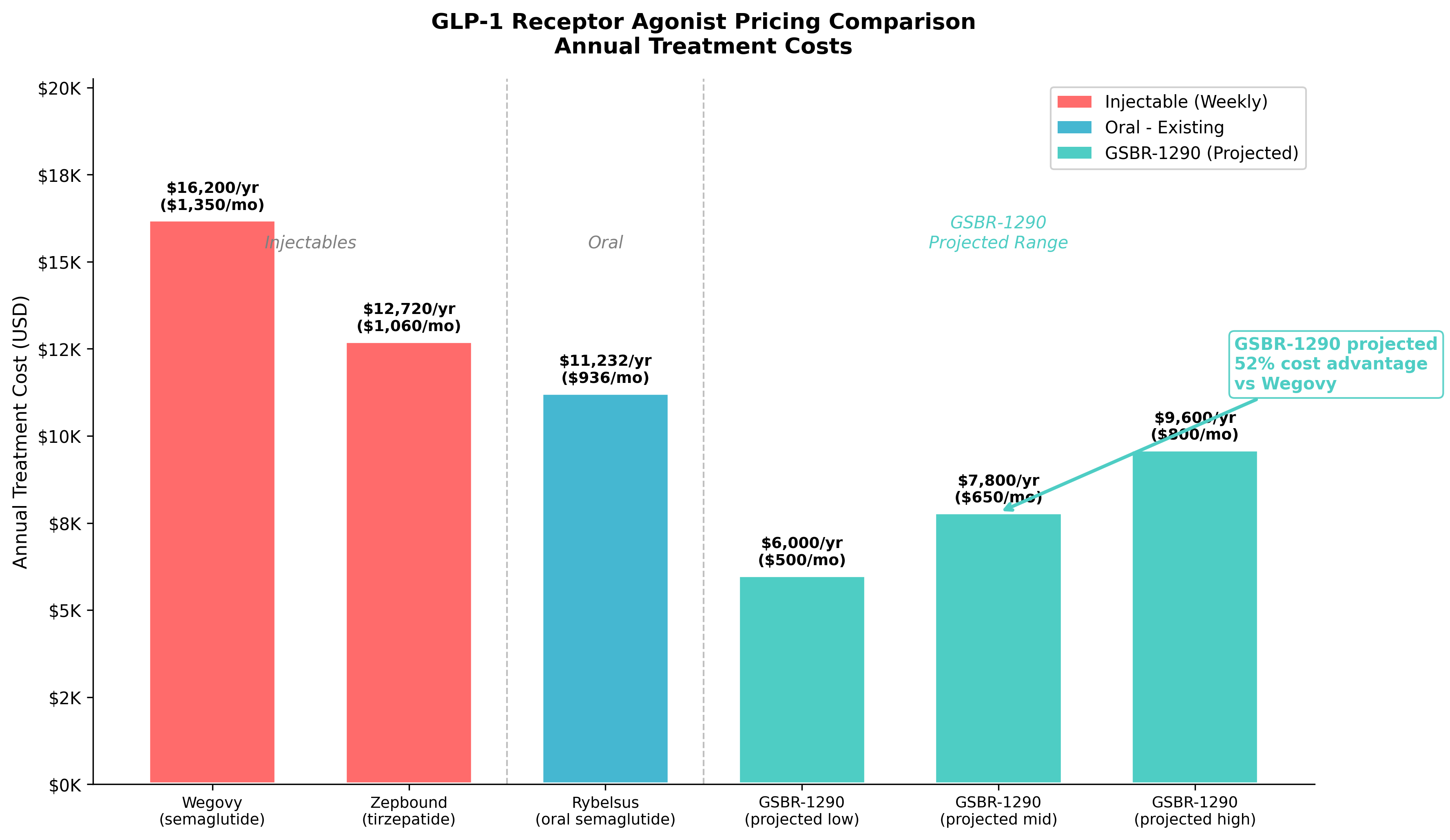 Figure 6: Annual pricing comparison. GSBR-1290 is projected at $6,000-9,600/year, representing a 30-50% discount to Wegovy.
Figure 6: Annual pricing comparison. GSBR-1290 is projected at $6,000-9,600/year, representing a 30-50% discount to Wegovy.
| Therapy | Annual Cost |
|---|---|
| Wegovy (semaglutide, injectable) | $16,200 |
| Zepbound (tirzepatide, injectable) | $12,720 |
| Rybelsus (semaglutide, oral daily) | $11,232 |
| GSBR-1290 (projected) | $6,000 - $9,600 |
A 30 to 50% pricing discount versus approved competitors matters a lot for payer access, particularly as pharmacy benefit managers face mounting pressure to control GLP-1 spending. Medicare Part D currently excludes anti-obesity medications, but that policy will shift at some point, and a lower price point positions GSBR-1290 well when it does.
IP & patent analysis: adequate protection for the runway
K-Dense Web compiled Structure Therapeutics' patent portfolio and benchmarked it against competitive filings across the GLP-1 landscape.
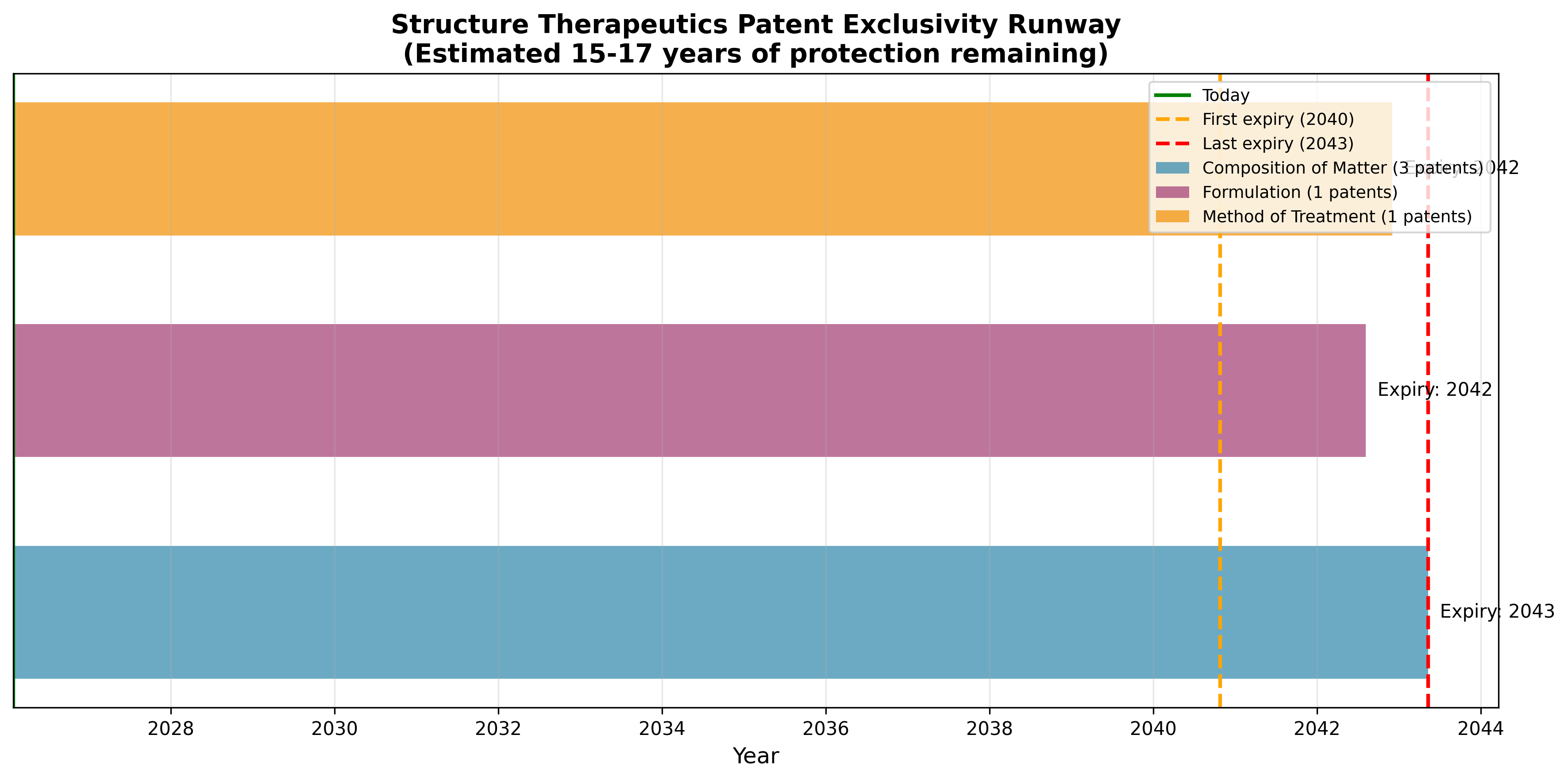 Figure 7: Patent exclusivity runway. Structure Therapeutics holds composition-of-matter patents extending to 2040-2043.
Figure 7: Patent exclusivity runway. Structure Therapeutics holds composition-of-matter patents extending to 2040-2043.
| Assessment Area | Status |
|---|---|
| Composition of matter patents | Strong (3 filings covering core scaffold) |
| Exclusivity runway | 14-17 years (expiring 2040-2043) |
| Freedom to operate | Low risk (distinct chemical class) |
| Portfolio size vs. competitors | Small but focused (5 families vs. 85+ for Eli Lilly) |
| Overall IP risk | Moderate-Low |
Composition-of-matter protection is the gold standard in pharma IP. Structure's portfolio is smaller than its big pharma peers, but it covers the core chemical scaffold with 14 to 17 years of exclusivity. For a Phase 2 asset, that's enough runway to protect the commercial opportunity through peak sales and well beyond.
Investment thesis: speculative buy with binary event risk
K-Dense Web synthesized all seven analytical steps into an investment thesis with SWOT analysis, Porter's Five Forces, and risk-adjusted NPV scenarios.
SWOT analysis
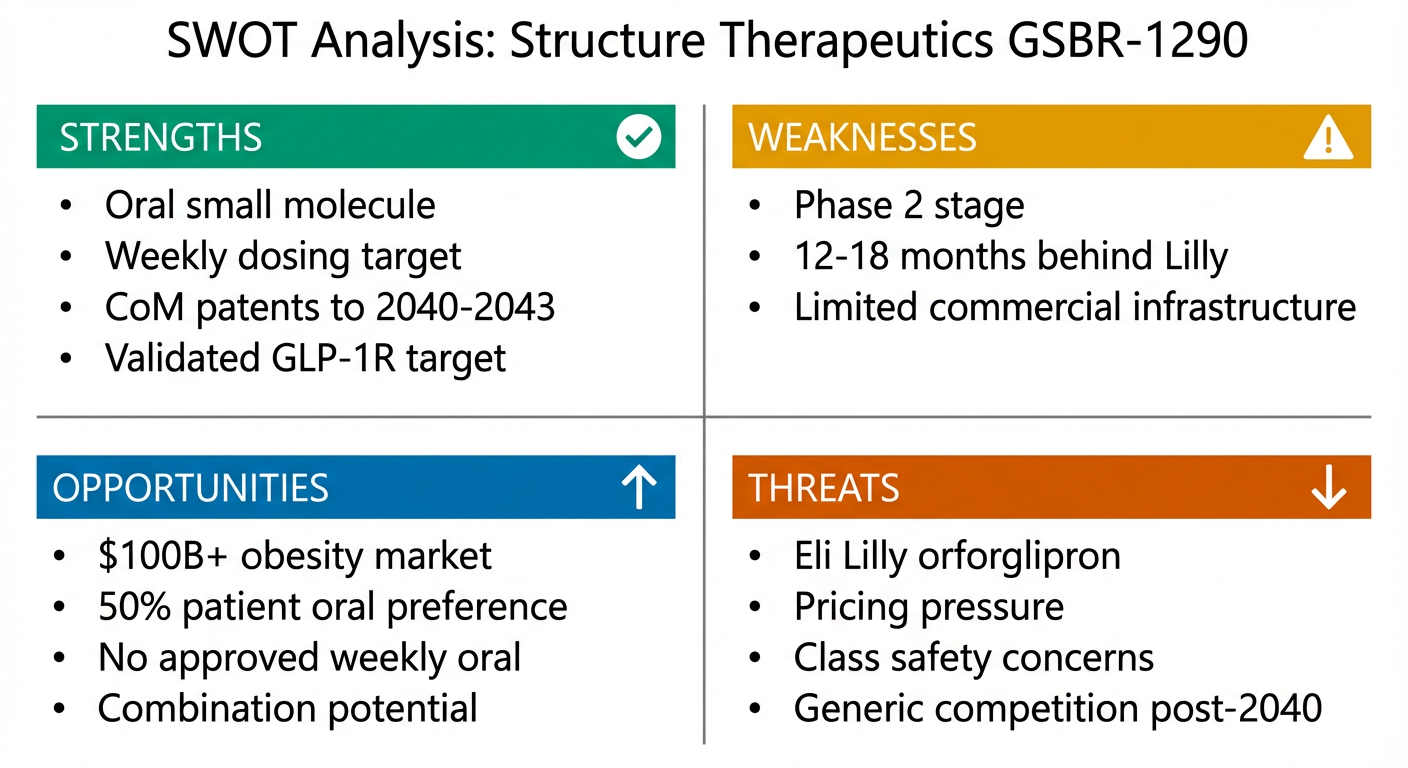 Figure 8: SWOT analysis. Strong market tailwinds and differentiated modality offset competitive and execution risks.
Figure 8: SWOT analysis. Strong market tailwinds and differentiated modality offset competitive and execution risks.
Porter's Five Forces
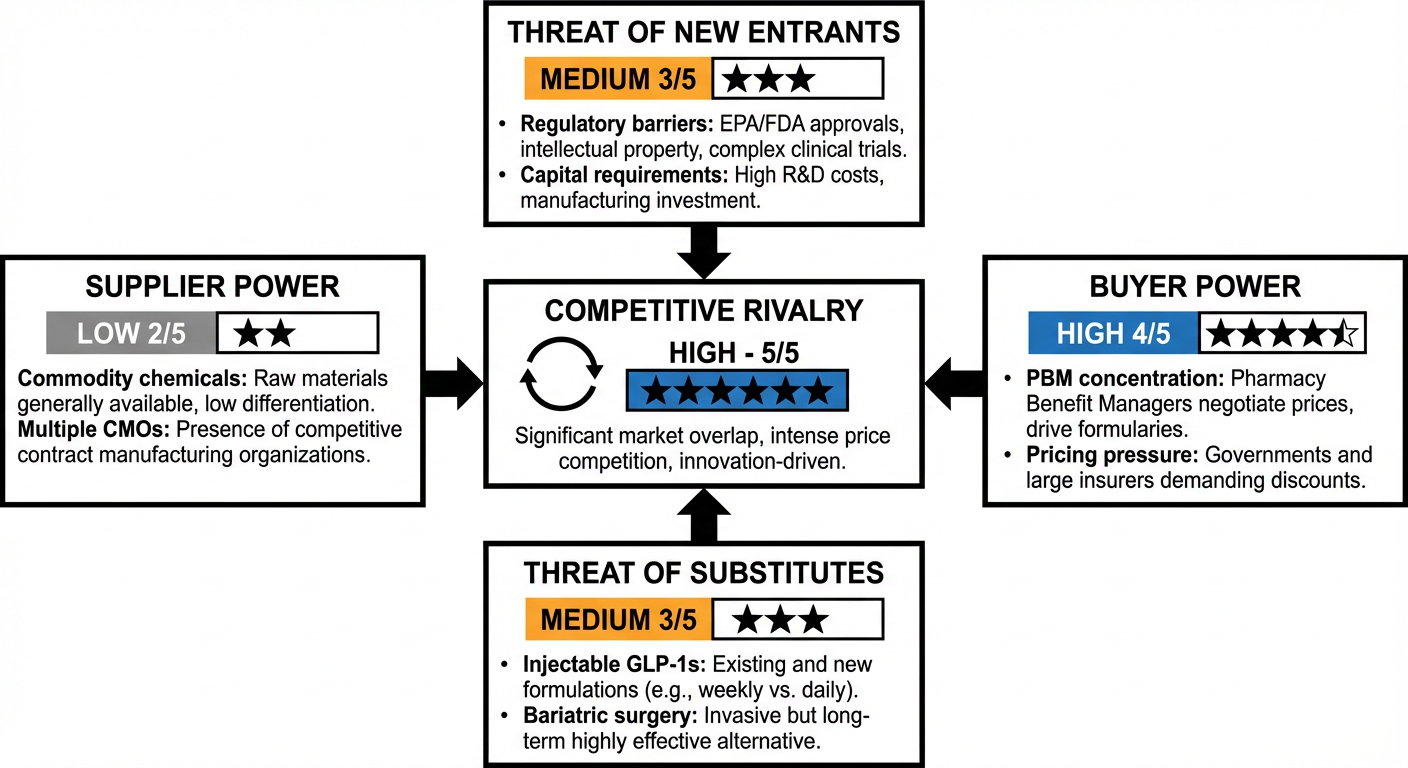 Figure 9: Porter's Five Forces. Buyer power and competitive rivalry are the dominant forces shaping the market.
Figure 9: Porter's Five Forces. Buyer power and competitive rivalry are the dominant forces shaping the market.
Buyer power (PBMs and payers) and competitive rivalry both score 4 to 5 out of 5. This is not a market you win with a mediocre product. Differentiation and pricing strategy are existential.
Risk assessment
K-Dense Web scored 20 risk factors across four categories:
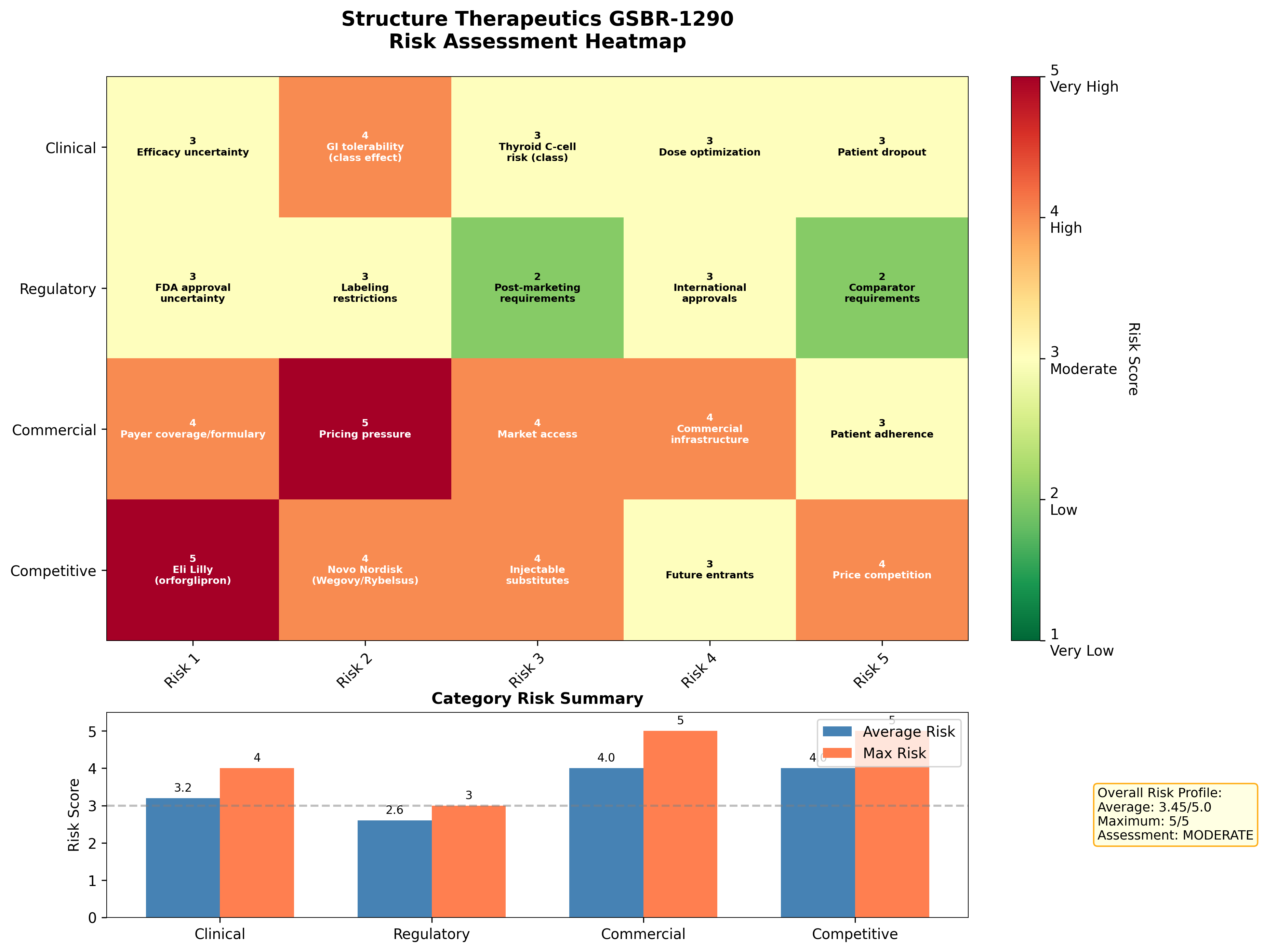 Figure 10: Risk heatmap. Commercial and competitive risks are the highest-scoring categories.
Figure 10: Risk heatmap. Commercial and competitive risks are the highest-scoring categories.
| Category | Average Score | Max Score | Assessment |
|---|---|---|---|
| Clinical | 3.2 / 5 | 4 | MODERATE |
| Regulatory | 2.6 / 5 | 3 | LOW-MODERATE |
| Commercial | 4.0 / 5 | 5 | HIGH |
| Competitive | 4.0 / 5 | 5 | HIGH |
Overall risk score: 3.45 / 5.0 (Moderate-High)
Valuation: risk-adjusted NPV of $2 billion
| Scenario | Peak Sales | Probability | Risk-Adjusted NPV |
|---|---|---|---|
| Bull Case | $10.0B | 25% | $4.2B |
| Base Case | $6.5B | 50% | $2.1B |
| Bear Case | $2.5B | 25% | $0.6B |
| Weighted | $2.0B |
Investment scorecard
| Factor | Score | Weight | Weighted Score |
|---|---|---|---|
| Market Opportunity | 9/10 | 25% | 2.25 |
| Differentiation | 7/10 | 20% | 1.40 |
| Competitive Position | 5/10 | 25% | 1.25 |
| IP Protection | 7/10 | 15% | 1.05 |
| Execution Capability | 6/10 | 15% | 0.90 |
| Total | 6.85 / 10 |
Rating: SPECULATIVE BUY
Structure Therapeutics offers exposure to the GLP-1 obesity market through a differentiated oral weekly approach. The addressable market is real and the modality differentiation is genuine, but Eli Lilly's competitive pressure and the binary Phase 2b data readout in H1 2026 demand disciplined position sizing.
Key catalysts to watch
| Event | Timeline | Impact |
|---|---|---|
| Phase 2b obesity topline data | H1 2026 | Binary event that determines thesis viability |
| Eli Lilly orforglipron Phase 3 readout | 2026 | Sets competitive benchmark for oral GLP-1 |
| Phase 3 initiation | H2 2026 | De-risks development timeline |
| Partnership / licensing announcement | 2026-2027 | Validates commercial potential |
The full analysis package
This post covers the highlights. The complete due diligence package goes substantially deeper. K-Dense Web generated:
- 26 structured data files (CSV, JSON, TXT) covering clinical trials, safety events, patent filings, market sizing, KOL networks, and more
- 15 publication-quality visualizations (PNG and PDF formats)
- 6 detailed analysis reports (competitive landscape, safety assessment, MOA differentiation, market analysis, IP assessment, investment thesis)
- 1 compiled 52-page PDF report with LaTeX typesetting, citations, and appendices
All analysis scripts are included for full reproducibility.
Download the Full PDF Report (52 pages)
Explore the Complete Session Data
Manual research equivalent: 60 to 110 analyst hours
For a researcher working without K-Dense Web, this would realistically be a 1.5 to 3 week project, depending on how much prior GLP-1 context they already had and how polished the final investment memo needed to be.
| Workstream | Manual Researcher Estimate | What Drives the Time |
|---|---|---|
| Source collection and normalization | 8-14 hours | Pulling data from ClinicalTrials.gov, FAERS, PubMed, Open Targets, USPTO/WIPO, SEC filings, and market datasets into comparable formats |
| Competitive landscape mapping | 8-12 hours | Cleaning trial records, classifying modalities, identifying direct and indirect competitors, and building positioning tables |
| Safety benchmarking | 10-18 hours | Downloading FAERS data, filtering GLP-1 reports, grouping adverse events, and checking whether the signal is interpretable |
| Market sizing and pricing model | 8-16 hours | Reconciling prevalence, treated population, adoption, pricing, payer access, and scenario assumptions |
| IP and patent review | 8-14 hours | Searching patent families, reading claims at a high level, mapping exclusivity runway, and flagging freedom-to-operate risk |
| Investment synthesis | 12-20 hours | Turning raw findings into a thesis, risk scorecard, valuation scenarios, SWOT, Porter's Five Forces, and IC-ready narrative |
| Charts, formatting, QA, and citations | 6-16 hours | Producing visualizations, checking calculations, formatting the report, and documenting methodology |
| Total | 60-110 hours | Roughly 8-14 full working days for one experienced analyst |
At a fully loaded analyst or consultant cost of $150-250/hour, the manual labor equivalent is roughly $9,000-27,500 before any paid database subscriptions, expert calls, legal review, or senior partner oversight. Adding specialist review can easily push the effective cost above $35,000-50,000 for a fund-quality diligence package.
These estimates assume:
- The researcher is already familiar with biotech investing, GLP-1 biology, and public regulatory data sources.
- The scope is limited to public data, not management interviews, physician surveys, payer checks, or proprietary prescription datasets.
- Patent work is a high-level investment screen, not a formal legal freedom-to-operate opinion.
- The output standard is an investment committee draft with reproducible charts and cited assumptions, not a fully banked diligence report.
- The researcher starts from a blank workspace and must collect, clean, analyze, visualize, and write up the findings manually.
K-Dense Web generated the same style of public-data diligence package in a single autonomous session, including the underlying scripts, structured files, visualizations, and compiled 52-page PDF.
Why this matters for your fund
Traditional biotech due diligence on an asset like GSBR-1290 can take an experienced analyst 60 to 110 hours to produce. Querying ClinicalTrials.gov, pulling FDA FAERS data, building market models, analyzing patent filings, synthesizing a thesis: each step requires domain expertise and manual effort.
K-Dense Web compresses this into a single autonomous session. The platform:
- Queries real data sources (not LLM hallucinations): Open Targets, PubMed, ClinicalTrials.gov, FDA FAERS, bioRxiv, USPTO
- Generates quantitative analysis with reproducible Python scripts
- Produces IC-ready deliverables: executive summaries, risk heatmaps, and compiled PDF reports
- Documents everything with full data provenance and methodology
Whether you're screening pipeline assets, preparing for an investment committee meeting, or building conviction on a position, K-Dense Web cuts the timeline from 1.5 to 3 weeks of manual analyst work to a single autonomous research session.
K-Dense Web uses pay-as-you-go pricing — sign up and run a full due diligence like the one above.
Have questions about using K-Dense Web for biotech investment research? Reach out at contact@k-dense.ai.
Disclaimer: This analysis was generated by an AI system and has not been independently validated. It is provided for informational and demonstration purposes only and does not constitute financial, investment, or medical advice. Always consult qualified professionals before making investment decisions.
